Journal of the Ceramic Society of Japan
編集委員からのお知らせ
[2024/1/16] 2024年1月16日14時以降の投稿を対象に早期公開を開始します。
[2024/3/1] 特集号を募集します。 Advanced Functional Materials and Devices (AFMD-2)new
2023 JCS-JAPAN 優秀論文賞
 |
Decomposition of 2-naphthol in water and its antibacterial and antiviral activities by LaMnO3 and LaCoO3 in the dark R. Kiribayashi, K. Sunada, Y. Mochizuki, T. Isobe, S. Matsushita, T. Nagai, H, Ishiguro, A Nakajima, J. Ceram. Soc. Jpn. 131, 117 (2023). |
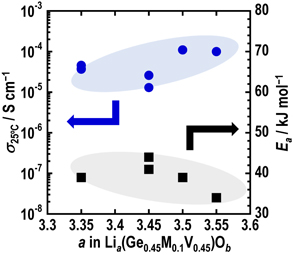 |
Preparation of Li4GeO4– Li3VO4 based electrolytes via mechanochemical treatment C. Okushima, Y. Yoneda, T. Kimura, K. Motohashi, A. Sakuda, M. Tatsumisago, A. Hayashi, J. Ceram. Soc. Jpn. 131, 141 (2023). |
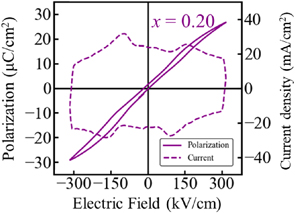 |
Effect of Ta substitution on the ferroelectric-antiferroelectric phase transition in tetragonal tungsten bronze-type K2NdNb5O15 H. Abe, S. Yasuhara, T.Tsurumi, T. Hoshina, J, Ceram. Soc. Jpn. 131, 189 (2023). |
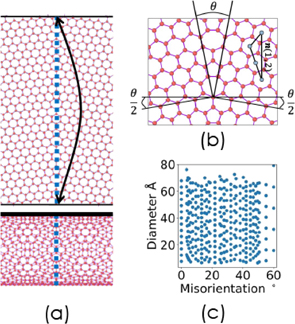 |
Tensile strength of a transverse grain boundary in a single-walled carbon nanotube Y. Xie, K. Shibata, T. Mizoguchi, J. Ceram. Soc. Jpn. 131, 621 (2023). |
2023 JCS-JAPAN 優秀総説賞
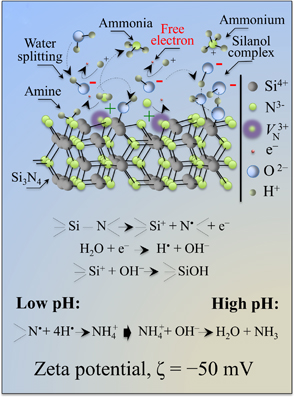 |
Silicon nitride as a biomaterial G. Pezzotti, J. Ceram. Soc. Jpn. 131, 398 (2023). |
2023 JCS-JAPAN 学生優秀論文賞
Most Cited
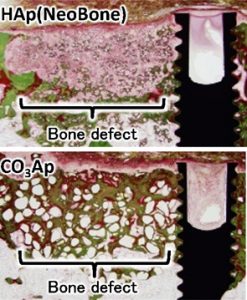 |
Carbonate apatite bone replacement: learn from the bone K. Ishikawa, J. Ceram. Soc. Jpn. 127, 595 (2019). |
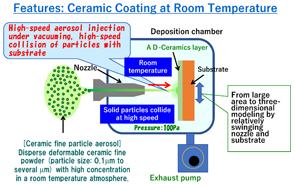 |
Room temperature impact consolidation and application to ceramic coatings: aerosol deposition method J. Akedo, J. Ceram. Soc. Jpn. 128, 101 (2020). |
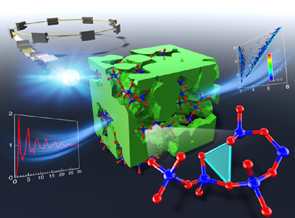 |
Understanding diffraction patterns of glassy, liquid and amorphous materials via persistent homology analyses Y. Onodera, S. Kohara, S. Tahara, A. Masuno, H. Inoue, M. Shiga, A. Hirata, K. Tsuchiya, Y. Hiraoka, I. Obayashi, K. Ohara, A. Mizuno and O. Sakata, J. Ceram. Soc. Jpn. 127, 853 (2019). |
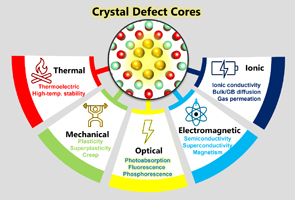 |
Ceramic science of crystal defect cores K. Matsunaga, M. Yoshiya, N. Shibata, H. Ohta and T. Mizoguchi, J. Ceram. Soc. Jpn. 130, 648 (2022). |
| Lithium-ion conductivity and crystal structure of garnet-type solid electrolyte Li7-xLa3Zr2-xTaxO12 using single-crystal K. Kataoka and J. Akimoto, J. Ceram. Soc. Jpn. 127, 521 (2019). |